TERMS
Epidermis = Outer most layer of the skin, serves as a protective barrier.
Dermis = Middle layer of the skin, actively growing and providing nutrients to the skin.
Sebaceous Glands = Glands at the base of hairs that release an oily substance to the surface to waterproof and protect the outer epidermis layer.
Eczema = Group of skin diseases featuring dry, red, itchy, oozing skin that may be caused by many factors; irritants (chemical or environmental), inflammation (Atopic Dermatitis), glandular disfunction, veinous disorders or neurological dysfunction.
Atopic Dermatitis = Group of inflammatory driven skin diseases, often identified by dry, red, itchy, oozing skin often in inner joint areas. This is a type of Eczema.
Psoriasis = Group of inflammatory skin diseases, often identified by areas of white/pink scaly and flaky plaques of skin often on outer joint surfaces. Can cause systemic inflammation.
Psoriatic Diseases = Term given to a class of more severe Psoriasis that involves other tissue such as joints, eyes and intestines.
Intro to the Skin
The term “Skin” most often refers to the outer layer of cells that cover and protect most of our body (and other mammals too). Though in reality the skin is a very complex organ that contains not just this outer barrier, but a complex network of cells that contribute to physically shielding us from injury, sensing environmental changes, regulating our body and comprising complex immune functionality that serves to both repair damage and ward off any invader that might break through. Unfortunately, there are times when this complex multi-faceted system becomes dysfunctional or is coopted, inducing disease. In this installment of Making Science Accessible I’ll be discussing the basics of skin anatomy (structure) and function, followed up with deeper dive into two major skin diseases, Atopic Dermatitis (common type of Eczema) and Psoriasis. Discussing how they’re similar, how they differ and why these relationships can alter how one seeks treatment and reprieve from the disease.
Most people see The Skin as a single layer of protection on the outside of our body, but it’s really much more complex than that. The skin is comprised of three major layers; outer Epidermis (that we see), Dermis (middle) and the underlying fatty layer. The Epidermis is what most people are familiar with, serving as the physical barrier to the outside world, protecting us from contact injury, UV damage (melanin) and infection. This outer layer is constantly being rebuilt by dividing keratinocyte (skin) cells that slowly push upwards until they reach the surface as a tightly woven network of dead cells, creating a solid barrier. The second layer, the Dermis, is the thickest part of the skin and is where most of the action happens. The Dermis contains most of the blood vessels that supply the skin with nutrients, lymph vessels that house our immune cells, hair follicles, nerve endings and sweat/oil glands. The innermost layer of the skin is the Subcutis, made up of a network of collagen and fat cells that serve to insulate and absorb shock, protecting the underlying tissue and organs. Dysfunctions can occur within any layer of the skin (cancer, structural, inflammatory), though this writing will primarily focus on immune related diseases that originate in the Dermis, subsequently affecting the surrounding tissue.
Eczema vs Psoriasis
The two most common forms of inflammatory skin disease are Eczema and Psoriasis. On the surface (drum roll) they would appear to be very similar, but once you dig deeper into their contributing disease mechanisms, there are noticeable differences. First, I’ll do a brief compare and contrast of the two, before diving into a more detailed discussion about each individual disease class, it’s contributing mechanisms, clinical signs and treatment options. Eczema is a broader term that is medically used to describe a group of different skin disorders that all manifest similarly. Eczema can be caused by underlying immune dysfunction (Atopic Dermatitis), contact with irritants such as chemicals and allergens (ie Poison Oak/Ivy), nerve irritation, defects in the Sebaceous glands (severe cases of dandruff) and vein disorders. While all of these can cause similar dry, red, itchy and oozing skin rashes, I’ll be focusing mostly on Atopic Dermatitis, the most common chronic form of Eczema.
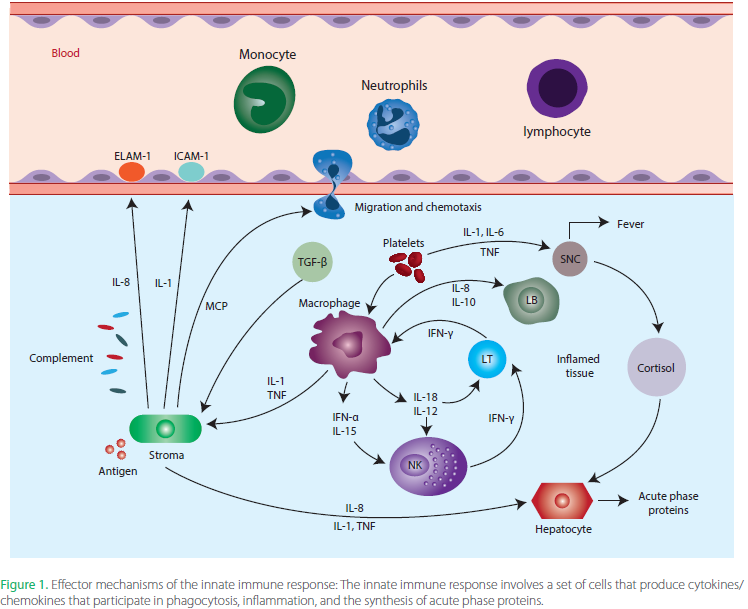
While both Atopic Dermatitis and Psoriasis lead to Epidermal and Dermal irritation and barrier damage, the specifics of their outward appearance are often what’s used for differential diagnosis. The differences in their external appearance are often caused by dysfunction of different immune and cellular mechanisms within the body, but there is also much overlap between the two diseases as well. Both can have genetic mutations that predispose one to developing the disease, and this linkage is typically stronger the more severe disease an individual suffers. Of the genes that are commonly dysregulated in both diseases, approximately 80% overlap, but that also means that 20% of what is dysfunctional is different. Both diseases are commonly driven by hyper-activated Tcells in the Dermis, leading to excessive inflammation, Epidermal barrier breakdown (ie skin damage) and overall dysfunction. But when one drills down at a more functional level, Atopic Dermatitis is primarily driven by Type 2 Tcells (Th2), while Psoriasis is driven by Th17/Th22 Tcells (that produce signaling molecules IL-17 and IL-22). These differences in signaling and function recruit different parts of the immune system to the skin, meaning that the primary cells involved in the inflammation and dysfunction can differ. Though the clinical reality is that both Atopic Dermatitis and Psoriasis exist on a spectrum, where both the clinical manifestations and the underlying cellular mechanisms can be quite variable from individual from individual. Meaning that while general trends in appearance and causes might exist, each case (patient) has its own unique fingerprint. In fact, there is growing evidence that some patients can suffer from both diseases, with them either occurring simultaneously or alternating (known as Psoriatic Dermatitis). In these cases, the immune dysfunction signatures and clinical signs will overlap or alternate, further confounding diagnosis and treatment. In the next few sections I will be discussing each disease in isolation, though you may notice a lot of overlap between the causes and treatments, further showing how complex and intertwined immune related diseases can be in general.
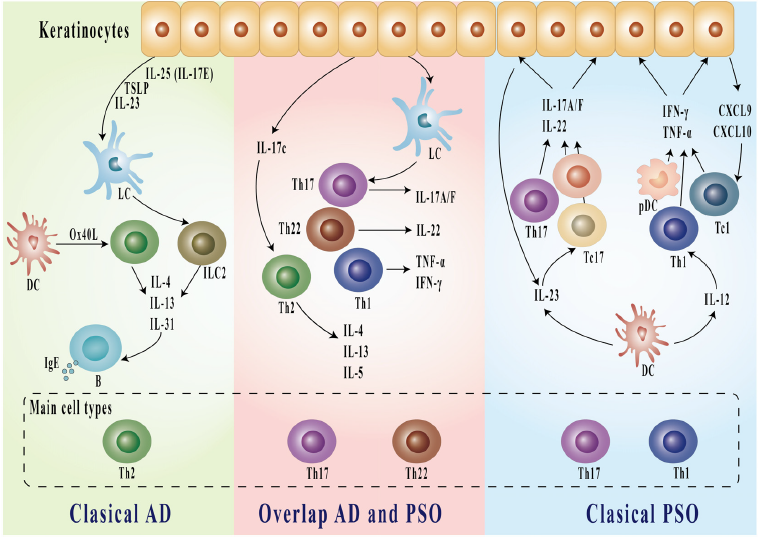
Figure 1, Li et al. Beyond the Dichotomy: Understanding the overlap between Atopic Dermatitis and Psoriasis.
Atopic Dermatitis/Eczema, Disease Biology, Variability and Treatment
While the term Eczema is often used interchangeably to describe Atopic Dermatitis (one form of Eczema), medically it’s a classification for a group of dermatitis skin disorders with similar clinical appearance, but different underlying mechanisms. Skin disorders such as Seborrheic Dermatitis (sebaceous gland dysfunction), Contact Dermatitis (chemical or allergic irritant induced) and other less common diseases all fall under the same Eczema umbrella. Seborrheic Dermatitis is associated with a dysfunction of sebaceous glands, which produce an oily substance that serves to lock in skin moisture and prevent infections. When these glands don’t produce the appropriate amount of fluid the skin can become irritated, dried out and/or prone to infection by fungi (commonly Malassezia spp). This dysregulation leads to inflammation and the dry itchy skin rashes commonly associated with Eczema. For Seborrheic Dermatitis, appearance wise this often manifests as more severe cases of dandruff or facial Eczema. Common treatments include anti-fungal and anti-inflammatory creams and shampoos to treat the condition, which is typically well managed. The Contact Dermatitis form of Eczema is commonly associated with skin rash formation driven by an allergic reaction in response to an external sensitivity, such as a chemical or allergen. Common forms of Contact Dermatitis are Poison Oak/Ivy reactions, reactions to detergents and other household chemicals. Treatment can be as simple as cleaning the affected area and letting the irritation calm down or include topical and oral anti-inflammatory therapies in more severe cases. As one who suffers from both mild Contact Dermatitis to chemical irritants and severe skin reactions to Poison Oak/Ivy I can attest to how painful and debilitating the condition can be, but also how well it’s managed with special cleaning agents and steroids.
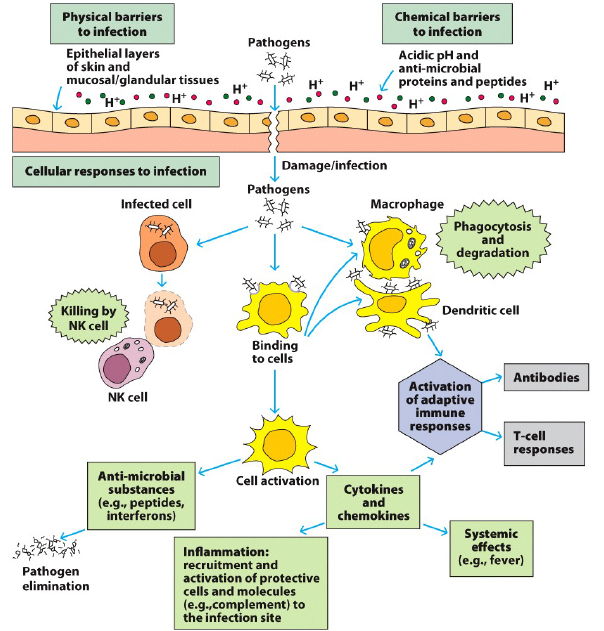
While Seborrheic and Contact Dermatitis are typically well controlled with treatment, Atopic Dermatitis (AD) can be a more problematic disease due to variability in the underlying causes and more inconsistency in treatment efficacy. Mutations in the skin structural proteins (filaggrin), Th2 signaling pathways (IL-4 and IL-13) and other immune activators have been linked to increased risk for developing AD. These are all linked to increased activity of the Th2 pathway, which is the primary driver of AD disease and skin damage. Th2 responses are directed by a specific subset of Tcells that are typically activated by allergens and parasites. When properly regulated, Th2 immune responses help to clear these external trouble-makers from our body by activating IgE Antibody production and Mast Cells and Eosinophil cells. These serve to wall off the foreign entities and then break them down to be removed from the skin. In AD these responses become inappropriately activated, and the cycle of overly exuberant Th2 mediated immune activity drives degradation of the epidermal barrier (skin), allowing additional allergens and bacteria (such as Staph aureus and Streptococcus, common in AD patients) to penetrate the skin, creating additional inflammation and damage that can become self-propagating. In some cases the skin damage comes first, in others the underlying immune activity, regardless the end result is a predominantly dysregulated Th2 immune response in the skin that drives continual activation, damage and irritation. While the dysregulated Th2 immune response is the most prominent immune feature in AD, high levels of activity in Th17 (IL-17 signaling Tcells) and Th22 (IL-22 signaling Tcells) cells can be contributors to disease. These Tcells use different signaling pathways to activate microbial defense mechanisms (Th17) or to activate skin proliferation and healing (Th22). Just as with the Th2 immune response, when properly regulated and activated in moderation they help protect and heal our body, but when over activated the Th17 pathway can cause additional tissue damage and the Th22 response can cause skin thickening and scarring, rather than normal healing. In summary, AD is typically caused when normal skin immune cell function becomes dysregulated and over-activated, which damages the Epidermis, causing irritation, which allows more foreign molecules to get under our skin, perpetuating the cycle of dysfunctional immune activation.
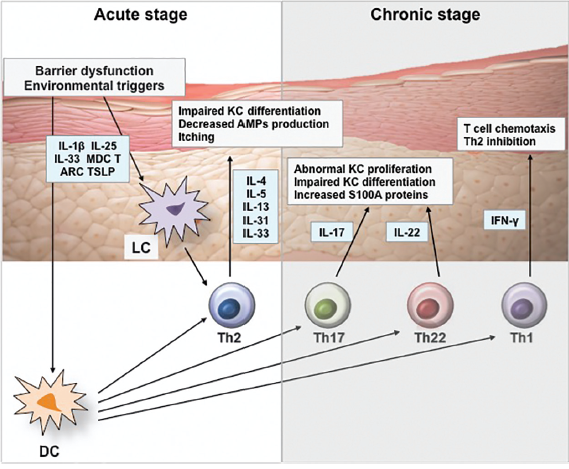
Figure 1, Kim et al. Pathophysiology of Atopic Dermatitis: Clinical Implications.
Because of the varied causes and contributing factors to AD and Eczema, optimal treatment for controlling skin damage and inflammation can vary depending on the individual case. Treatments come in four main categories: generic skin supportive therapies, topical anti-microbials, topical anti-inflammatory treatment and systemic anti-inflammatory treatment. For more mild forms of AD and Eczema basic topical treatments to clean, hydrate and support the skin can help mitigate some of the itching and irritation, though this is more of a supportive than specific disease targeting treatment. These can include lotions/creams to moisturize the skin, UV/sun exposure and regular cleaning of the affected area. Antibiotics and antifungals (mostly topical) can also be helpful to mitigate some of the potential infection risk associated with certain bacteria and fungi, reducing the infection induced immune activation. While these won’t treat the underlying immune dysfunction, they can help reduce severity and immune activation. The next classification of treatments, topical anti-inflammatories, are effective for many cases of AD/Eczema in addition to other skin diseases (like Psoriasis). Broad-spectrum topical anti-inflammatories include common compounds such as corticosteroids, cyclosporin and methotrexate, all of which serve to generally suppress immune responses, with no specificity for cell type or pathway. This broad-spectrum efficacy is both their strength and their weakness. It’s a strength because they can work against any underlying immune dysfunction (where overactivation occurs) regardless of the pathway. So even if one doesn’t know the specific immune dysfunction, these broad-spectrum immunosuppressants can help. The weakness is, because they don’t target only the dysfunctional immune pathways, other protective systems in the body can also be suppressed AND the dysfunctional pathway can even be suppressed too much, leading to susceptibility to other infections. These are common risks and side effects of taking broad-spectrum anti-inflammatories for prolonged durations and is why they aren’t the ideal therapeutics for long term maintenance of a disease, but can be helpful to reduce short term flares or acute disease.
The last category of treatments for AD/Eczema are systemic anti-inflammatory treatments. While this can include broad acting anti-inflammatories like those mentioned above, for AD/Eczema systemic treatments usually leverage cell type or pathway specific therapies. Things such as monoclonal antibodies and drugs that are specifically designed to bind to one cell, receptor or signaling molecule. These types of targeted therapeutics can be great because treatment can target only the dysfunctional parts of the immune system without reducing the properly functioning areas. Treatments exist to target IL-4 and IL-13 (Th2 pathway), IL-17 and IL-23 (Th17 pathway), IL-12/23 (Tcell activation) and JAK/STAT (generic immune activation), all of which have been shown to help people suffering from AD/Eczema depending on the immune mechanisms that are dysfunctional. The challenge with these therapeutics is the variability of AD/Eczema disease in each person and the challenge of identifying the exact mechanisms of dysfunction for a single individual. Each targeted treatment will help some individuals more than others, and while there is testing that can help identify these pathways, it’s not highly utilized and readily available to most patients. I’ll discuss this more in the next post about “Personalized Medicine and Immune Signatures”.
Psoriasis and Psoriatic Arthritis, Disease Biology, Variability and Treatment
Like Eczema (and Atopic Dermatitis), Psoriasis is a skin disease characterized by Epidermal damage, irritation and underlying immune dysregulation, but with some striking differences in both its external and internal manifestation. The cellular hallmark of Psoriasis is keratinocyte dysfunction (these are the cells that make up our skin), primarily caused by the underlying immune dysfunction and hyper-activity. This immune dysfunction is usually dominated by activation of Dendritic Cells (immune activators) that cause excessive activation of Tcells of the Th17 (activate IL-17 pathways) and Th22 (activate IL-22 pathways) subtypes. Helper Tcells then recruit additional inflammatory mediators, especially neutrophils (cells that destroy invaders and clean up damaged cells), that further perpetuate a cycle of inflammation induced tissue damage and tissue damage induced inflammation, and round and round we go. These pathways in turn alter the growth and maturation of keratinocytes (skin cells), damaging skin integrity and causing Epidermal irritation. So, while Eczema diseases can be driven by an external component (ie infection/irritants), Psoriatic diseases tend to be driven by the underlying inflammation, making external factors a secondary symptom.
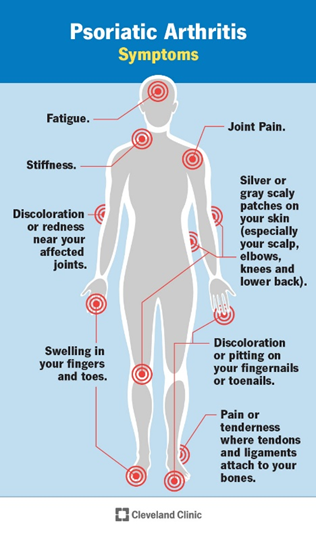
It’s also not uncommon for the underlying immune disorder that causes Psoriasis to also cause additional systemic disease in other organs such as joint arthritis, inflammatory bowel disease, cardiovascular disease and other metabolic diseases because the inflammation doesn’t always stay localized just to the skin, especially in more severe cases (20-30% of cases). Recently the term “Psoriatic Disease” has been used to describe these more complex cases, driven by similar immune dysfunction, but not being localized to just the skin. Of these, joint arthritis (inflammation) is the most common escalation of skin Psoriasis, leading to a condition known as Psoriatic Arthritis. Most people are familiar with the term Arthritis, which generically refers to inflammation or irritation of the joint tissue (any joint). Psoriatic Arthritis specifically is a chronic disease that links skin Psoriasis immune dysfunction to inflammation in the joint tissue, causing swelling, irritation, tissue damage and potentially bone loss in severe cases. This can be especially debilitating, occurring in the fingers and toes (Distal interphalangeal), in multiple joints all over the body (Symmetric or Asymmetric), in the spine (Spondylitis) or in the hands and feet (Mutilans). The joint inflammation occurs in two categories, non-specific activation (unregulated cellular infiltration and inflammation mediated by signaling molecules and Neutrophils) and tissue targeting auto-immune disease (Tcells, Bcells and Antibodies that directly target joint tissue). Both can cause similar symptoms and damage, but the cell types and molecules involved can differ, leading to differential treatment approaches. A growing body of evidence suggests there are numerous gene mutations that increase the likelihood of one developing Psoriasis, especially HLA genes (involved in recognizing self vs foreign molecules), IL-12/IL-23 immune pathway as well as mutations affecting IL-17 and IL-22 Tcells. These discoveries support the rationale for targeting these pathways in treatment to help mitigate disease severity in patients suffering with Psoriatic Disease. The difficulty arises in identify which of the many immune mechanisms is driving the dysfunction, how out of balance they are in each patient and how much are other associated immune components involved (such as Th1 Tcells, Bcells, Natural Killer cells and gamma-delta Tcells). This variability in disease severity AND involved cell types is what drives the differences in how a patient is treated and what patients are treated with.
Because Psoriatic Diseases often include a more systemic component than Eczema, many preferred treatments target the specific immune pathways mentioned above systemically. I’ll break down the treatments into four categories; generic skin supportive therapies, topical anti-inflammatories, systemic corticosteroids and Disease-modifying antirheumatic drugs (DMARDs) that aim to slow down the immune response. Measures that promote overall systemic and skin health can help mitigate less severe Psoriasis symptoms, such as a balanced diet, skin moisturizing, short term UV/sun exposure and regular cleaning of affected areas. For Psoriasis that is primarily skin associated with no major systemic involvement, topical anti-inflammatory medications (both steroids and non-steroidal) can provide temporary relief to the irritation and skin damage during flares, though they aren’t typically recommended for long-term use. In moderate to severe cases, systemic treatments can provide better long-term remission in many patients, the challenge lies in which treatment is best for an individual due to the variability of disease. Again, systemic corticosteroid use can help to rapidly reduce inflammation but also serves to broadly suppress total immune function and as such is not recommended for long-term use. Other Disease-modifying antirheumatic drugs (DMARDs) such as methotrexate and cyclosporin can help to reduce systemic over-activation of the immune system, but since they are broad acting and not immune pathway specific they can also lead to immunosuppression and a reduced ability to fight off other diseases in some patients. In recent years new classes of immune pathway specific therapeutics have been developed to target the parts of the immune system that are commonly dysregulated in Psoriatic Diseases including; TNFa signaling molecules, IL-12/23 pathway, IL-17 Tcell pathway, JAK/STAT/Tyk2 (broader immune activating molecules) and Bcell/Antibody production. If you watch TV you’ve probably seen commercials for Skyrizi/Tremfya (IL-23 blockers) or Otezla (PDE4 blocker) recently, these are all newly approved medications for Psoriasis and related diseases. These types of treatments are designed to specifically block and reduce activity in a specific immune pathway in an attempt to try and bring the immune system back into balance, rather than broadly suppressing the whole immune system. While specificity can be very beneficial, the two challenges are identifying WHAT the right pathway to target is and mitigating the side-effects of blocking that immune pathway and other treatment associated side-effects. Unfortunately, in many cases identifying the most effective treatment with the fewest side-effects can be a challenging process, wrought with trial and error, changes in medication and evolving disease symptoms. This becomes a very personalized process that needs to be addressed on a case-by-case basis.
Concluding Remarks
Many of us know people suffering from some form of skin disease, with Eczema and Psoriasis being the two most common. While they share some common symptoms and treatments at a more basic level, they are driven by a wide range of different cell and immune dysfunctions that can require much more specific attention as the disease severity worsens. Treatment can range from basic cleaning and skin hydration to short-term anti-inflammatory treatments to long-term immune modifying therapies. And while these different treatments can help reduce symptoms and mitigate life altering challenges, management often becomes a life-long process for many suffering from these diseases. For long-term immune modifying treatments, the process of optimizing treatment can be challenging and frustrating. Whenever one attempts to modify an immune response, the body naturally creates a feedback loop and response to the treatment, and while for many these feedback loops to the treatment are minimal, sometimes this can create new challenges or immune dysregulation that needs to be understood and addressed. This process is confounded by the fact that currently our healthcare system doesn’t tend to prioritize personalized immune function testing to help identify an individual’s personal immune dysfunction and immune signature, slowing down the process of finding the optimal treatment. In the next posting I’ll be tackling the topic of “Personalized Medicine and Immune Signatures”, where the technology stands to making this a reality, how it might help guide an individual’s treatment, help us understand our own body function, but also where the challenges lie in understanding and applying the information gained. While there can be a lot of power in gathering individual personalized data, a single test rarely tells the whole story and with our ever evolving understanding of the human body there are still many gaps in this knowledge.
This content is for educational and informational purposes only and is not a substitute for medical advice. It does not provide diagnosis, treatment recommendations, or guidance for any individual’s specific medical situation.
References:
• Armario-Hita JC et al. Updated Review on Treatment of Atopic Dermatitis. J Investig Allergol Clinn Immunology. 2023; 33(3): pg158-167.
• Brunner P et al. The Immunology of AD and its Reversibility with Broad Spectrum and Targeted Therapies. J Allergy Clin Immuno. April 2017; 139 (4 Suppl): pgS65-S76.
• Cleveland Clinic. Psoriatic Arthritis. https://my.clevelandclinic.org/health/diseases/13286-psoriatic-arthritis. 2025.
• Dall’Oglio F et al. An Overview of the Diagnosis and Management of Seborrheic Dermatitis. Clin, Cosmet and Invest Dermatology. Aug 2022; 15: pg1537-1548.
• El-Esawy FM et al. Methotrexate Mechanism of Action in Plaque Psoriasis: Something New in the Old View. J Clin Aesthet Dermatol. Aug 2022; 15(8): pg42-46.
• Emmungil H et al. Autoimmunity in Psoriatic Arthritis: Pathophysiological and Clinical Aspects. Turkish Journal of Med Science. Aug 2021; 51: pg1601-1614.
• Freitas E at al. Bimekizumab: The New Drug in the Biologics Armamentarium for Psoriasis. Drugs in Context. 2021; 10: 2021-4-1.
• Gossec L et al. EULAR Recommendations for the Management of Psoriatic Arthritis with Pharmacological Therapies: 2023 Update. Ann Rheum Disease. March 2024; 83: pg706-719.
• Kim J et al. Pathophysiology of Atopic Dermatitis: Clinical Implications. Allergy and Asthma Proceedings. March 2019; 40(2): pg84-92.
• Li M et al. Beyond the Dichotomy: Understanding the Overlap Between Atopic Dermatitis and Psoriasis. Front Immunology. Feb 2025; 16:1541776.
• Loiselle AR et al. Prevalence and Co-Occurrence of Eczema Types in Adults in the United States: Insights from the All of Us Research Program. J of Invest Dermatology. March 2025; 145: pg2360-2363.
• Lowes M et al. Immunology of Psoriasis. Annu Rev Immunol. 2014; 32: pg227-255.
• NIH National Cancer Institute SEERS Training Module. Anatomy of the Skin. https://training.seer.cancer.gov/melanoma/anatomy/. 2025.
• Orlando E, Nona L. Eczema Vs. Atopic Dermatitis: Causes, Diagnosis, Treatment. University of Central Florida. https://ucfhealth.com/our-services/dermatology/eczema-vs-atopic-dermatitis/#:~:text=Eczema%20and%20atopic%20dermatitis%20(AD),out%20of%20seven%20different%20types. 2025.