The Immune System is a complex interplay between a dozen+ cell types, signaling molecules, anti-microbial systems and physical barriers that serves to protect our body from internal and external invaders in addition to leading clean up and repair of natural damage that occurs as part of daily life. The goal of this writing isn’t to outline every detail and nuance of the Immune System, but rather to provide a general overview of the major pieces and parts that contribute to protecting our bodies, allowing us to navigate life in a complex world full of dangers. In addition to providing a few relatable real-world examples of how our immune system protects us, I will also include some information on how it can become dysfunctional in its own right, leading to self-inflicted harm. This article will be a precursor to many more detailed and disease specific articles focused on creating comprehensible and digestible understanding of complex disease topics that affect life.
TERMS:
Innate Immune System = Non-specific pieces of the immune system.
Adaptive Immune System = Pathogen specific immune system.
Mucosa = Soft tissue that lines organs such as the airways, intestines and reproductive tract, allowing regulated entry/absorption from the surroundings to the rest of the body.
Pathogen = Agents such as viruses, bacteria and parasites that can infect a person.
Antigen = Any material foreign (from the outside world) or naturally occurring in the body that can cause an immune response.
Anti-microbial = Something that is known to stop the growth of or kill microorganisms such as bacteria, viruses, fungi or parasites.
The Immune System is divided up into two separate arms, the Innate Immune System which is made up of quick reacting non-pathogen specific protection mechanisms and the Adaptive Immune System, comprised of antigen specific responses that can be both short lived and very long lived (memory). The Innate Immune System is made up of three arms; natural barriers to entry (such as skin and airways), circulating molecules that destroy invaders and damaged cells and immune cells that wander the body looking for things to clean up. All of these mechanisms function in a pathogen agnostic manner, meaning it doesn’t matter if the invader is Bacteria A or B, Virus A or B. Meanwhile the Adaptive Immune Response exists for when these initial mechanisms get overwhelmed and our body needs to create a more specific response in order to eliminate the threat before it can cause too much ancillary damage to our own body. T-cells (Helpers and Killers) and B-cells (producing Antibodies) are the two main components of the Adaptive System that get amplified in response to a specific target. Once the threat is cleared, a portion of these cells can remain dormant in our body for years, just waiting for the same target to return (our Immune Memory). If that’s all you are interested in learning about the specific cells and pieces of the Immune System, feel free to skip down to the sections on “Health, Immune Function and Dysfunction” below, as the next section is a more detailed and denser explanation of the Immune System’s parts and pieces (for the nerds out there).
Components of the Innate and Adaptive Immune System
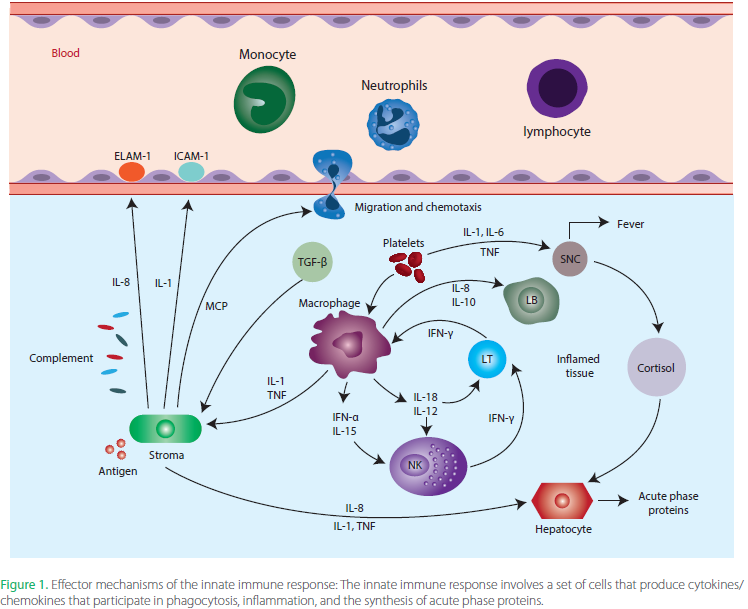
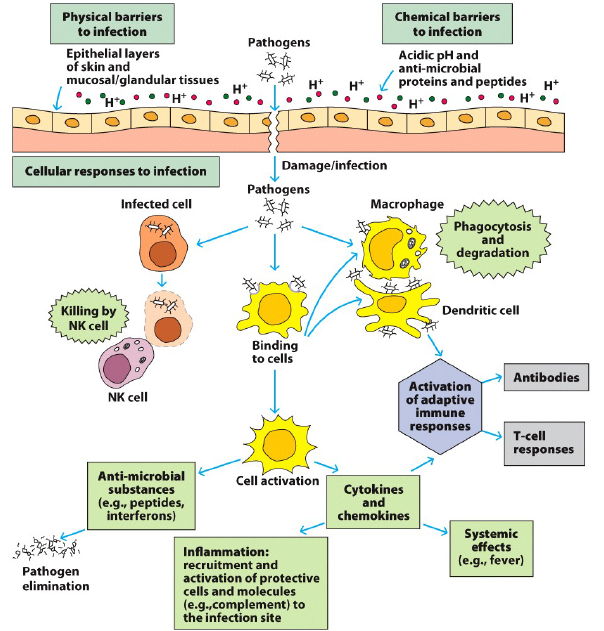
Immune exposure and development begins in utero, but really kicks into full swing after birth, when we are thrust into a dirty world full of new antigens and pathogens. While the process of exposure and reaction to our environment never stops after birth, the human Immune System is mostly formed by 3-4years of age. The first line of defense of the Innate Immune System are natural barriers like the skin and mucosal cells lining the airways, intestines and reproductive tract. These create physical barriers that provide limited and restricted access to the body, helping to prevent bacteria, viruses and allergens from regularly gaining entry. In addition, hair in the nostrils along with mucus in the airways and intestines help trap outside materials, allowing the cells that line these surfaces to produce natural anti-microbial molecules that are capable of destroying some pathogens. If these external barriers and systems are damaged or overcome, then the body has two additional sets of quick responding Innate mechanisms that serve to clean up and protect our organs and tissues. The first of these are a series of circulating molecules that include anti-microbial peptides (similar to natural antibiotics), proteins designed to tag and identify foreign molecules (eg CRP, lectins and ficolins) and complexes that help destroy infected or damaged cells (complement proteins). None of these are living, but all are capable of interacting with their targets to assist in protecting our body.
The second set of Innate mechanisms comprise a diverse group of cells that are both programmed to both dispose of garbage and to signal other more specialized cells to enhance the immune response. All of these cells have Pattern Recognition Receptors that are designed to identify common patterns found on bacteria, viruses, parasites, environmental allergens and damaged cells. These common antigens are things that our bodies are exposed to daily, and that we’ve evolved to recognize and clear out as part of our normal immune function. Granulocytes are some of the first cells to respond to issues, these include cells such as; neutrophils that help eat up foreign invaders and destroy damaged cells, eosinophils that help kill parasites and mast cells and basophils that are commonly involved in responses to allergens. The next class of cells collectively known as phagocytes (Monocytes, Macrophages and Dendritic Cells) serve two important functions, to eat up and destroy any garbage they find floating around and in more extreme cases to use this garbage to activate more specific Adaptive Immune responses (remember this in the next section). The last subset of Innate Immune cells are specially programmed killers, whose sole goal is to destroy foreign invaders and mutated cells (ie cancer cells). They are responsible for maintaining the balance between normal growth and dysfunction (parasites, mutations and cancers), preventing the latter from coopting our body.

While the Innate Immune System has some generic patterns it recognizes, it is NOT responding in a pathogen specific way. Meaning it would respond to most bacteria using similar mechanisms and the same goes for many similar viruses. If the first lines of defense fail to contain the pathogen/damage, cells known as Antigen Presenting Cells (often Phagocytes) are able to trigger activation of the pathogen specific Adaptive Immune Response. The two cells many people are familiar with that drive Adaptive responses are T-cells and B-cells. There are three major classes of T-cells, each serving a different role in enhancing specific immune responses; CD4+ T-helper cells, CD8+ Cytotoxic T-cells (specialize in killing) and gamma-delta T-cells (Immune surveillance). During normal development, each CD4+ and CD8+ T-cell is created with a unique random target, in fact there are millions of these unique cells just floating around the body. If by chance one of the Antigen Presenting Cells finds a new infection that matches the unique target on one of these T-cells, then that unique cell multiplies in response to the specific activation signals. The CD4+ Helper T-cells role is to identify the location of these unique infections and to signal other killer cells and B-cells to migrate to the site of infection/damage, thus specifically amplifying the immune response. There are Helper T-cell types that target infections hiding inside our cells, others that target free floating infections, some that participate specifically in allergic responses, others that help in tissue repair and even some that are specifically programmed to slow down inflammation if things get too wild and crazy. Meanwhile the CD8+ Cytotoxic (Killer) T-cells have overlapping specificity to the Helper T-cells, but instead of recruiting other cells and amplifying immune response, their sole job is to kill any cell that matches their unique target. This includes both infected cells and mutated/cancerous cells.
The last arm of the Adaptive Immune Response is the infamous B-cell and the Antibodies they produce. Just like the T-cell, during normal times our body creates an army of millions of B-cells, each with a unique random target, just hanging around, waiting for a Phagocyte or T-Helper cell to show them the right target. If they find the unique antigen that matches their Antibody, they go into action, multiplying and cranking out Antibodies against the target. These Antibodies are basically very sticky keys (though shaped more like a Y), that bind to the infection/target, both marking it to be destroyed by the killer cells (discussed above) and walling off the target to prevent it from doing further damage to the body. In cases where the body amplifies T-cells and B-cells against a very specific infection/target, some of those cells mature and turn into memory cells that go into a hibernation like state, hanging out for years (or even our entire life). These cells lie in wait for that same infection/target to return, ready to quickly respond and prevent us from getting sick (this is generally how vaccines work). In total, our Immune System is a complex web or responses and interactions, all tightly regulated with feedback loops to allow careful responses to invading pathogens, repair damaged tissues and prevent excessive collateral damage to our own body from within.
Health, Immune Function and Dysfunction
You might be asking, how do all these cells and pathways affect how my daily bodily function, or when I get sick with a bacteria/virus, how about when things become dysfunctional leading to self-inflicted disease? Even more than the details above describing the different parts of the Immune System, these questions are much more pertinent to life on a day-to-day basis. Though unfortunately in some ways these questions are far more complicated than the Immune System itself. My goal in this section is to introduce some general trends and concepts, rather than to address every health condition, supplement, disease, Autoimmune condition out there. In subsequent posts I’ll be addressing specific diseases, conditions and health topics that are either of specific interest to me or that have been requested by people in my life (feel free to reach out with your request).
The Immune System is something that’s constantly working, reacting, resting and repairing the body every single day. The surrounding environment is full of agents that have the potential to harm us, cause disease or coopt our cellular machinery, and the Immune System is one of the major factors that keeps a lot of these in check. This includes preventing allergens from entering the body, killing off bacteria/viruses that we’re exposed to on a daily basis to repairing damaged tissue from all the cuts and scrapes we accumulate, the Immune System does a little bit of everything. Our external environment is constantly challenging our body’s defenses, and for the most part it does a good job of blocking out many things, killing off small numbers of invading pathogens, clearing out cancerous/dead cells and repairing both internal and external tissue damage. There are many aspects of our daily lives that help keep things functioning well, including proper nutrition, a good supply of energy (aka calories), adequate sleep, low stress and exercise. Even if we did everything perfectly to keep the body functioning at 100% (basically impossible), our body’s defenses and repair mechanisms sometimes need a little extra help.
The most common challenge our Immune System faces is an overwhelm by too many pathogens. If a pathogen is either too numerous or has mechanisms to evade the immune system, then our body starts going into overdrive, manifesting in what most of us think of as ‘getting sick’. Our body temperature increases (fever) to both inhibit pathogen replication and increase immune activity, our vessels flood immune cells to the site of infection (causing inflammation), we have aches and pains (caused by stimulation of local nerves and the battle that’s being waged around them) and we become fatigued as our body reallocates energy to fighting off the infection. This infection can happen locally around a cut/wound, in a specific tissue (like a stomach bug or respiratory infection) or systemically (in your blood) affecting the whole body. There are varied degrees of severity of this process, at it’s best our body responds quickly to control the invading pathogen and we barely feel any symptoms (vaccines help prime these responses to be quicker), but at it’s worst the invading pathogen spreads throughout our body both infecting and killing cells directly but also hyper-activating our immune system so much so that it causes self-inflicted damage in its effort to clear the infection (collateral damage). So in general it is good to let the body go through the inflammation and healing process naturally, though if a fever gets too high or the immune response too severe, it can start to damage organs and cause more harm than good, and that’s when it’s a good idea to seek medical attention (especially for younger children). Unfortunately, sometimes it’s hard to know when our body crosses this line and what the best line of care is, because how each of us responds and how much each individual can tolerate is different.
The other challenge our immune system regularly faces is trying to keep the body from dysfunctionIng and directly attacking itself. This can occur in two main forms, Autoimmune disease (where the body mistakenly attacks itself) and Cancer (where mutations cause uncontrolled growth). Autoimmune diseases cover a wide-range of issues including Rheumatoid Arthritis, Type 1 Diabetes, Eczema and Psoriasis, Crohn’s disease, Multiple Sclerosis and Lupus among many others. In all of these diseases, some part of the Immune System becomes misdirected and hyperresponsive, leading to the body attacking itself and damaging a specific tissue or cell type. These diseases can be triggered by genetic factors, infections that cause a misfiring of the Immune System, random mutations causing auto-reactive cells, environmental triggers, chemical damage and other factors that we’ve yet to identify. Because of the varied causes of Autoimmune Diseases, we can’t always predict who or when they might occur. Even more challenging is that not all Autoimmune Diseases of a single classification are created equal, meaning one person with Eczema (skin disease) might have a different pattern of immune dysfunction than another person, so what treatments and interventions work best of each could be different (coming in the next blog). These are the types of discussions I’ll be trying to dive deeper into as part of future writings, discussing the more subtle nuance of a single disease or classes of diseases and what that means for those working through those challenges.
The other major classification of dysfunction is the diverse field of Cancer Biology. In over-simplified terms, Cancer is caused by a mutation that occurs somewhere in the body (really could be anywhere) that creates cells that divide, grow and spread in an uncontrolled manner. These cells can be in Immune Cells, epithelial cells, structural cells, nerve cells and so on. Regardless of the cell type, during normal maintenance processes, the Immune System is responsible for identifying these malfunctioning cells and destroying them before they can divide and spread, but when they are missed during the normal surveillance process is when disease can happen. These mutations can lead to the growth of either benign or malignant tumors, the former being a non-cancerous clump of cells (don’t spread and invade other tissues) and the latter growing rapidly and spreading throughout the body causing serious risk to normal tissue function. While there are some patterns in the mutations/cells that cause Cancer, there is also a lot of variability, which adds to the challenge of treating the disease, especially in the context of minimizing damage to non-cancerous tissue in the body, not always an easy feat. As such there is a wide range of therapeutic options, ranging from targeted radiation that kills the cancerous cells (and nearby cells), drugs that can interrupt cell division and growth, specialized therapies that can tag cancer cells for the immune system to identify (if they are unique enough) and the use of specially designed synthetic immune cells specially designed to attack the cancerous cells (one of the newer technologies). While the Oncology field has made great strides in understanding and treating Cancer, there are still many cancers that are hard to predict, identify and treat, meaning much more research is needed to improve the understanding of the what, how and why.
The process of healthy and normal immune function is a complex web of maintenance, reaction and regulation. Serving to protect our bodies, maintain normal growth and fix damage. All of the information above is just a small window into the complexities of disease and immune functionality. It’s also important to keep in mind that we’re all individuals and while I’ve outlined some general patterns that apply to most humans, we each have our own unique challenges, strengths and body types that need to be treated as such. This blog series is intended to be an ever-evolving work that serves to help those with Non-Scientific backgrounds further understand complex health related topics. As such I welcome all questions and any constructive comments and critiques from Scientist and Non-Scientist alike. Up next, “Skin Diseases, Disruption and Immune Dysregulation”…. If you’re a more visual learner, check out the University of California Televisions video on Immunology Basics, it’s about 1.5h long, so strap in.
This content is for educational and informational purposes only and is not a substitute for medical advice. It does not provide diagnosis, treatment recommendations, or guidance for any individual’s specific medical situation.
References
- Abbas A et al. Cellular and Molecular Immunology. Elsevier Science, 2003.
- Alberts B et al. The Cell. Garland Science, Taylor and Francis Group. March 2002.
- Alotiby A. Immunology of Stress: A Review Article. Journal of Clinical Medicine, 2024; 13, pg6394.
- Anaya JM et al. Autoimmunity, From Bench to Bedside. El Rosario University Press. September 2013.
- Goldsby R et al. Kuby Immunology. WH Freeman & Company. January 2002.
- Jain N. The Early Life Education of the Immune System; Moms, Microbes and (missed) Opportunities. Gut Microbes, Sept 2020.
- Shishido SN et al. Humoral Innate Immune Response and Disease. Clinical Immunology, June 2012; 144, pg142-158.
- Wrotek S et al. Let Fever do it Job. Evolution, Medicine and Public Health, Nov 2021; pg26-35.