Precision Medicine vs One-size-fits-all:
The typical One-size-fits-all medical diagnostic practice in the US aims to use examination of symptoms and broad more generic testing to put an individual’s health situation into any number of predefined bins for treatment and management. While this system can be efficient in dealing with more general and routine health issues, it often lacks the personalized nuance required to break down more complex medical situations or to optimize an individual’s treatment plan to their specific needs. Enter Precision or Personalized Medicine… When many people hear the terms Precision or Personalized Medicine, they often think of medicines that are specifically made for an individual person, but that’s not typically how the Medical community uses these terms. In the Medical community, the term Personalized Medicine (used going forward to include Precision Medicine) refers to creating a medical management plan for an individual, taking into account their personal medical history, genomics, individual biology, environment and lifestyle information. The primary goal of Personalized Medicine is to create treatment plans for a person based on their personal situation, rather than using the more traditional One-size-fits-all medical treatment binning. On the surface it sounds like a fantastic way to improve our healthcare and get each person the detailed and individualized treatment they need to manage the ever-growing complexities of our lives, but there are numerous hurdles and limitations currently holding back Personalized Medicine from being implemented more broadly. In the following paragraphs I’ll first outline some of the types of testing and information that could be included as part of the Personalized Medicine process, then outline what aspects are currently being used in the Cancer and Autoimmune spaces. Following that up with a discussion of what testing and diagnostics could be implemented to vastly improve treatment in these two disease spaces. Lastly, wrapping up the discussion talking about the major hurdles blocking implementation and some potential ethical issues that need to be resolved as part of the process. I recommend reading through my “Intro to Immunology” post prior to starting into this writing to better understand some of the general terminology and how some of this testing ties into Immune Responses and Functionality.
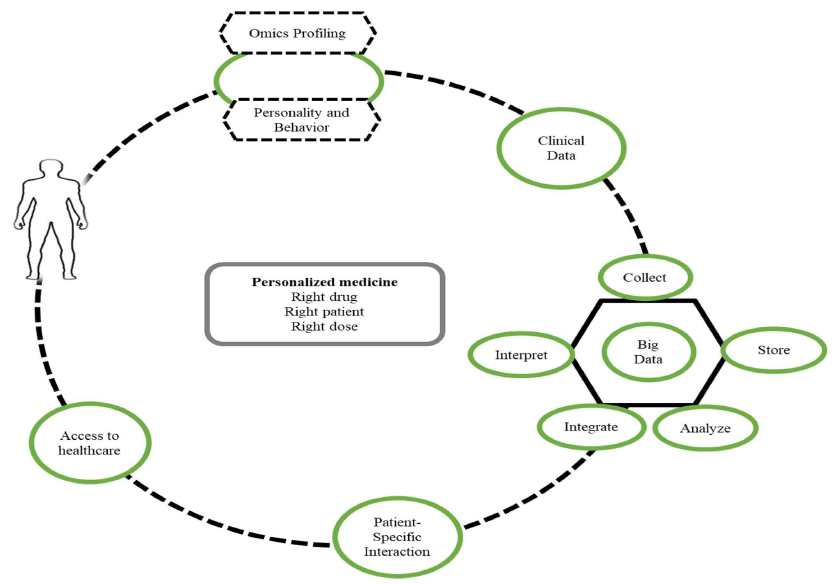
The Different Pieces of Personalized Medicine
The idea of Personalized Medicine doesn’t just consider the symptoms an individual is experiencing, but also multiple other more personal factors that can lead to a more nuanced and well-rounded understanding of an individual’s personal medical situation. More general things like personal family history, environmental factors (where you live, what you’re exposed to, diet, etc), social environment and general lifestyle are several types of Personalized information that are often collected as part of an individual’s personal medical file in the US, but they aren’t always considered to be important when selecting treatments and disease diagnosis. Some examples of things that ARE often taken into consideration are; Family history when it comes to hereditary diseases, environmental items like allergen exposures, toxin/chemical exposures for some cancers, diet for IBD/IBS and lifestyle when it comes to cardiovascular/heart issues. Beyond these very broad concepts, which can be helpful in understanding disease context, there are numerous more specific Personalized metrics that can help us understand each individual health situation more directly. The most prominent of these are genetic testing, proteomic testing and immune function profiling. In the next several paragraphs I’ll discuss each one in a little more detail, what they are, how they could drive understanding of a few representative diseases and how prevalent they are in our current Personalized Medicine environment. If you’re not as interested in learning about the specifics of the types of testing and instead are more interested in their implementation within personalized healthcare, skip down to the “Current State and Where It’s Going” section.
The most well-known form of Personalized Medicine testing is probably genetic testing. As many people might be aware, our genes (made up of segments of DNA that are barcodes for most things in our body) drive much of how our body functions, responds and its ability to fix itself. Genetic testing can fall into three categories when it comes to diseases; testing for baseline gene mutations to identify predispositions to specific diseases, testing of malignant or malfunctioning cells in the body that are causing disease, and testing for how an individual’s body will react, process and breakdown drugs (Pharmacogenomics). The first type is what many people might be more familiar with, testing our DNA composition to see what genetic sequences exist that COULD provide a pathway to developing disease at some point. Some common gene mutations associated with diseases occur in BRCA1/2 and HER2 (breast cancer), BRAF (melanoma), PIK3CA (many cancer types), HLA-B27 (Autoimmune Diseases), CFTR (cystic fibrosis), HBB (sickle cell), HTT (Huntington’s) and so on. There are THOUSANDS of genes that have been associated with different diseases, and while some genetic mutations can directly cause a disease most only link to an increased risk of MAYBE developing the disease in the future, but this is not diagnostic or guarantee any specific outcome. All these genes exist in each and every one of us, it’s when there is a mutation that changes how the gene functions that creates the predisposition to developing a certain disease, in most cases simply having the gene is a normal thing. The second type of genetic testing is diagnostic when disease has already developed. Many types of cancers and Autoimmune diseases are driven by dysfunctional genes within the malfunctioning cells, some of these are pre-existing and some can be caused by mutations that occur later in life. Some of these mutations can be identified by genetic testing to help Doctors and Scientist understand The What and Why of an individual’s disease. The last type of genetic testing is known as Pharmacogenomics, which encompasses looking at how specific genes affect drug binding, travel within the body, break down and processing. So rather than focusing on how our own body is functioning, this arm of genetic testing is focused specifically on how drugs interact and affect an individual. Pharmacogenomics can help guide drug selection, the amount dosed (depending how fast a drug is metabolized by an individual) and prevent giving medications that a patient might be likely to have an adverse reaction to. All these factors are affected by natural variations (and some aberrant mutations) in our genes that also affect the function of different systems within the body. Every year we (as a Scientific/Medical community) learn more and more about which genes and mutations are associated with predisposition to various diseases, functional pathways, resistance to other diseases, the ability to react to certain environmental conditions and so forth. Every one of us has a unique genetic makeup that drives many aspects of our health and functioning, but this is far from the only contributor to understanding our Personal health situations.
Proteome analysis is the next type of testing that can be utilized to help understand and direct Personalized Medicine. In short, each gene is a set of instructions to create a protein, these become the functional building blocks that make up most of our body. Both the sequence of these proteins (based on their genetic instructions) and the amount of each protein present within the body matter. The field of Proteomics seeks to understand if there are mutations in the design of the proteins and/or if there are changes in how much of a given protein are being produced. This can be done in a broad shotgun approach (looking at 100s or 1000s of proteins at once) or honing in on one specific protein to be used as a Biomarker of a certain disease. The term Biomarker is often used to describe a protein (or other marker) that can be measured and associated with the presence (or absence) of a specific disease. Some common protein Biomarkers that people might be familiar with are; CRP for inflammation, PSA for Prostate cancer, Troponin for cardiac issues, HbA1c for diabetes, Amyloid-beta and Tau for Alzheimer’s. Beyond this there are 100s of other markers that could potentially be tested that have shown to be associated with various diseases and have the potential to help direct therapeutic intervention and identify diseases before disease escalates.
The last area of Personalized profiling I wanted to discuss is Immune Function Profiling. As previously discussed in my “Intro to Immunology” post, the immune system is involved not just in fighting off infections but plays a key role in cleaning up damage and debris within our body and maintaining functional homeostasis. There are numerous different ways to analyze and understand how an individual’s immune system is functioning. One way is by counting the number of each immune cell type in the body (Complete Blood Count, CBC) to assess how many of each a person has. Another is by using protein Biomarkers (described above) to track various proteins associated with the activation of different immune pathways. In more complex cases there are even laboratory tests that can be run to assess the exact functional capacity of the immune cells that exist within an individual. All of these can help shape the picture of how well an individual’s immune system is functioning, where deficiencies or dysfunctions might be and what pathways and cells might be the most appropriate to target in the case of specific disease diagnoses. With the invention of Biologics (therapies that mimic something naturally occurring in the body) and Cell Therapies (injecting cells to directly fight a disease, such as CAR-T), the ability to identify the exact cell type, pathway and molecules within the immune system that are dysregulated or being over-produced can be critical in selecting the right treatment for an individual’s disease. The power of genetic testing, proteome analysis and Immune profiling lies in the ability to gather critical information about an individual’s personal biological makeup and function, which can drive more specific diagnoses and personalized treatment. Unfortunately, there are still several hurdles to us (in the US) fully realizing this level of Personalized Medicine and replacing our One-sized-fits-all approach to treatment and diagnosis.
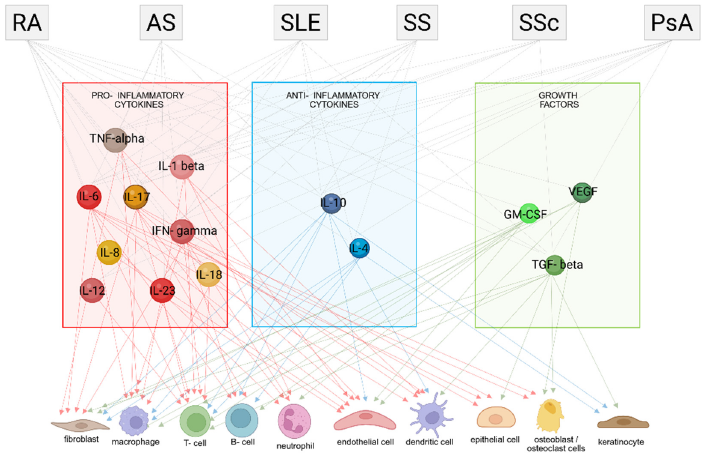
Current State and Where It’s Going
When it comes to the current (April 2026) impact Personalized Medicine has on an individual’s healthcare there are three different tiers; what’s widely available to everyone, what’s available in more specialized circumstances and what is available at the Research level but rarely or never utilized in the clinic. The current level of Personalized Medicine that’s widely available to most people on a prophylactic basis (prevention of diseases) is minimal compared to its potential. Genetic testing for numerous heritable diseases and genetic abnormalities is becoming more common-place as part of the prenatal care process and when a family member presents with a heritable disease. Even with this, there are 1000s of genetic tests that are rarely run outside of specialty situations where an existing disease is present but hasn’t been fully qualified by other testing or categorization. Genetic testing is most advanced in the field of Oncology (cancer treatment), where there is a wide array of testing that some cancer cells can be run through to help understand the cancer driving mutations, mutations that might create resistance to treatment and mutations that might drive metastasis (spread). While some testing like this does exist for Autoimmune diseases and other aging related diseases it’s less commonly used (see below for reasoning), instead treatment often progresses down the One-size-fits-all pathway, running patients through a checklist of 1st/2nd/3rd line therapeutics in a trial-and-error fashion.
When it comes to proteome analysis, there are a handful of tests that are widely used for tracking individual protein Biomarkers that map to different diseases and general health conditions (CRP for inflammation, PSA for Prostate cancer, Troponin for heart attack, HbA1c for diabetes, Amyloid-beta and Tau for Alzheimer’s to name a few), but this is just scratching the surface of what is possible in this field. Most of these tests exist as part of routine blood panels that Drs order through companies like Quest and Lab Corp, but rarely is broad shotgun proteomics analysis employed. Like the proteomics field, the broader implementation of Immune Functional assessment at the clinical level is very much lacking. CBCs (Complete Blood Counts) and CRP/ESR levels are really the only markers commonly assessed as part of diagnostic blood panels, outside of specialty situations. In specific disease cases there are a battery of different immune marker tests at clinician’s disposal; Antibody testing, specific cell type enumeration, microscopic analysis of biopsies of diseased tissue and genetic testing for immune system mutations. The trouble is that many of these tests don’t get to the root of many immune problems, only scratching the surface of the potential to understand an individual’s immune function. With the rise in Biologic therapies and cell therapies that are specially designed to target specific parts of the immune system, the ability to measure these targets and identify when they are dysregulated is the key to properly pairing a therapy with a disease. Unfortunately, while these tests (for proteins like IFNy, IL-17, TNFa, IL-1, CD20+ Bcells, Tcell subtypes, etc) do exist in the context of clinical trials, research/teaching hospitals and Research studies, they are rarely used for more general diagnostics. Instead, many clinicians tend to default to the One-size-fits-all approach and a trial-and-error process for determining which medication might work best for the patient. While there have been many advancements in regard to developing reliable Biomarkers and tests to track the development of many major diseases, there are still a few major hurdles preventing the implementation of what is both already available at the Research level and what is in development.
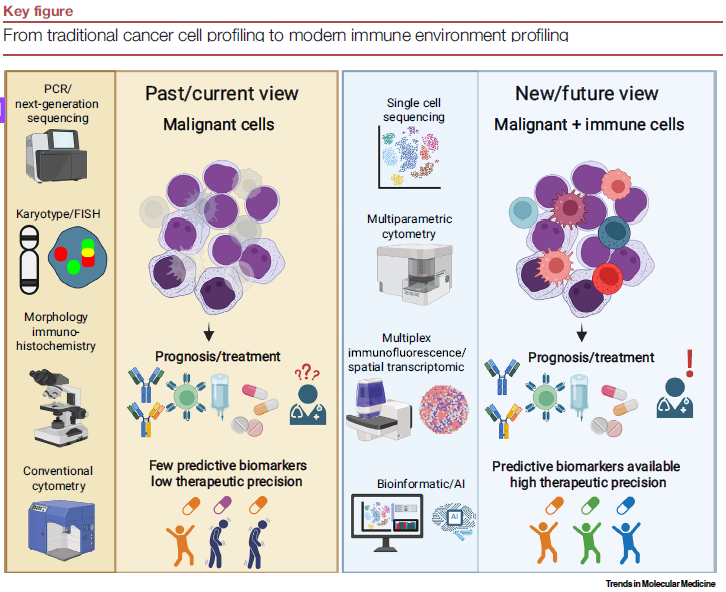
Right now (April 2026), what we as a Scientific community can do in the context of specialized clinical trials, in research/teaching hospitals and in the Research Laboratory setting exponentially dwarfs what is being utilized regularly in the clinics to help diagnose and treat patients. From detailed genetic mapping studies to understand the complex nuance of a single person, to in depth proteomic analysis tracking 1000s of important proteins that drive many aspects of human health, to complex immune function profiling that allows specific understanding of the strengths, weaknesses and dysfunctions of an individual’s immune system. Already there is so much more information and testing that could be utilized to help drive more Personalized health planning and treatment, but there are three main hurdles: test and data complexity, testing consistency and interpretation, and cost of implementation (time and monetary).
Things like detailed genetic and proteomic mapping generate huge amounts of complex data often requiring a very specialized multistep process to collate, perform quality control, summarize then interpret in a very nuanced manner. There is also the challenge that many of the genetic markers we’ve identified are not definitively diagnostic but instead contributors to disease potential (not certainty). Even then, it often requires multiple disease markers in combination to create a real risk, since many of the genetically linked diseases we deal with are multifactorial. In addition to the complexity issue, many of the Research Laboratory tests that currently exist for examining proteins and Immune function are not direct equivalents. Unlike genetic testing where we are often able to obtain a yes or no answer (in regard to mutation presence), protein and cellular analysis exists along a very large spectrum. Protein, cytokine (Immune cell signaling molecules) and immune cell functional tests can have a wide range of results with not all tests being of equal sensitivity and specificity. Tests from two different labs can be very rigorously designed to be highly consistent and accurate for detecting increases in a specific protein within themselves but comparing them to each other would not be appropriate. Think of it like trying to compare how one Teacher grades an essay to how another teacher grades the same essay, the scale could be shifted up or down depending on the system used even if good was still good. In addition to this, protein and Immune function levels can fluctuate from day to day, week to week and month to month in any ONE person, meaning that a single measurement isn’t always helpful in understanding an individual’s health situation. The real value lies in knowing if a test result deviates from our own personal baseline. The last major hurdle is the time and monetary cost associated with implementing a lot of these tests. While many are very feasible to perform, they take a lot more time than many of the routine tests which are efficiently automated, cost a lot more in supplies and personnel resources and would dramatically increase upfront insurance costs (though there’s an argument around long-term savings). So there is a cost-benefit to obtaining the depth of Personalized Medical information discussed above, it can be very costly and take lots of time to acquire, analyze and interpret and even then might not provide definitive answers.
For most of us this high level of detailed Personalized Medical information isn’t necessary to live a generally healthy life, but there is the question of how much more could our own personal health situations be optimized, diseases prevented or prophylactically treated and misdiagnoses prevented if each of us had all of this data as part of our routine health screening process? Ethically there is also the question of medical privacy and how to protect, maintain and secure all the data that comes along with this type of Personalized Medicine. Currently many companies performing genetic screening and complex disease analysis have the right to keep and use the data in what’s known as a ‘deidentified’ manner, meaning that it can’t be traced back to any one individual person. But what happens when we have so much genetic, proteomic and immune function data on an individual that it basically becomes a Biological fingerprint, unique to its owner? By understanding everything about our own personal Biology are we opening a brand-new type of discrimination and potential invasion of privacy if the data isn’t managed properly? As the world of Personalized Medicine continues to grow, these are just a few of the ethical questions that need to be discussed, in addition to the Scientific and practicality ones. If you are struggling from a complex disease or health situation that might benefit from additional Personalized testing that you are unable to get through more traditional providers and PCP (Primary Care Physicians), there are numerous teaching/research hospitals around the country (like CU Anschutz, Mayo Clinic, Duke, UPenn) that might be able to assist. Check out the Personalized Medicine Coalition for other clinics and locations that focus on advancing the use of Personalized Medicine in clinical patient care. Stay tuned for the next article “Microbiome, it’s Relationship to Digestion and Health”.
This content is for educational and informational purposes only and is not a substitute for medical advice. It does not provide diagnosis, treatment recommendations, or guidance for any individual’s specific medical situation.
References:
- Balch B. Making Medicine Personal: Moving Away From a One-Size-Fits-All Approach to Health Care. AAMC News. Feb 2024; https://www.aamc.org/news/making-medicine-personal-moving-away-one-size-fits-all-approach-health-care
- Biery N et al. Need for Standardization of Cytokine Profiling in CAR-Tcell Therapy. Molecular Therapy. Sept 2024; 32(9): pg2979-2983.
- Center for Disease Control. Pharmacogenomics. CDC, Genomics and Health. Nov 2024; http://cdc.gov/genomics-and-health/pharmacogenomics/index.html
- Delpierre C et al. Precision and Personalized Medicine: What Their Current Definition Says and Silences About the Model of Health They Promote. Implication for the Development of Personalized Health. Frontiers in Sociology. Feb 2023; doi: 10.3389/fsoc.2023.1112159
- Donniacuo A et al. Comprehensive Profiling of Cytokines and Growth Factors: Pathogenic Roles and Clinical Applications in Autoimmune Diseases. Int J Mol Sci. 2025; 26(8921).
- Gallo P. Cytokine Testing: Asking the Right Questions. J Allergy Clin Immunology. 2025; 156(6): pg1776-1777.
- Kay C. Immune Cell ‘Signatures’ Could Help Guide Treatment for Critically Ill Patients. Stanford Medical Center. Sept 2025; https://med.stanford.edu/news/all-news/2025/09/immune-cell-signature.html
- Knight V et al. Cytokine Testing and Challenges for Diagnostic and Clinical Monitoring Use. J Allergy Clin Immunology. 2025; 155(2): pg410-413.
- Krawczyk P et al. New Genetic Technologies in Diagnosis and Treatment of Cancer of Unknown Primary. Cancers. July 2022; 14(3429).
- Liesenfeld O et al. Clinical Validation of an AI-based Blood Testing Device for Diagnosis and Prognosis of Acute Infection and Sepsis. Nature Medicine. Sept 2025; 31: pg4044-4054.
- National Cancer Institute. Genetic Testing for Inherited Cancer Risk. April 2024; https://www.cancer.gov/about-cancer/causes-prevention/genetics/genetic-testing-fact-sheet
- Nygren P. Precision Cancer Medicine 2025: Some Concerns. ACTA Oncologica. Sept 2025; 64: pg1202-1204.
- Personalized Medicine Coalition. https://www.personalizedmedicinecoalition.org/
- Seremet T et al. Immune Modules to Guide Diagnosis and Personalized Treatment of Inflammatory Skin Diseases. Nature Communications. Nov 2024; 15(10688).
- Singh D et al. Personalized Medicine: An Alternative for Cancer Treatment. Cancer Treatment and Research Communications. Dec 2024; 42(100860).
- Stroe O. Proteomics: A Different Lens for Precision Medicine. European Molecular Biology Laboratory (EMBL). July 2024; https://www.embl.org/news/science-technology/proteomics-a-different-lens-for-precision-medicine/
- Sulieman L et al. The Use of Precision Medicine to Support the Precision of Clinical Decisions in Care Delivery. IMIA Yearbook of Medical Informatics. April 2024; pg 168-174.
- Unger S et al. Next-Generation Immune Profiling Beyond Blood Cancer Cells. Trends in Molecular Medicine. Dec 2025; 31(12): pg1140-1153.
